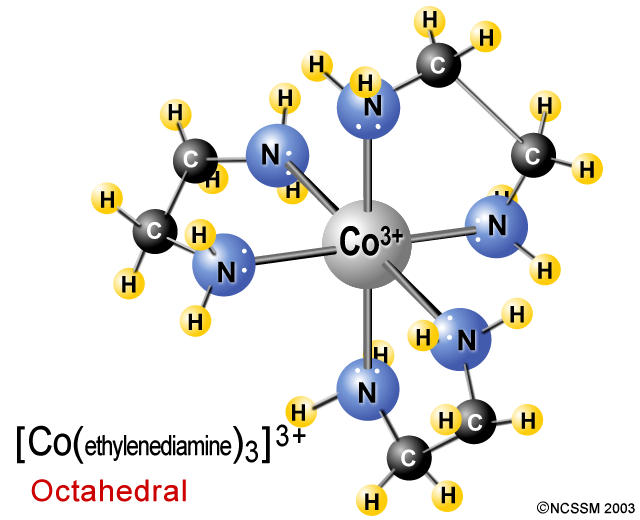
This gives it many useful applications! For example, it can be used to cure metal poisoning. It forms a complex by surrounding the metal ion. You can see from its shape that EDTA 4- is also a chelating agent. Multidentate ligand, A2 chemistry for AQA The EDTA 4- ion is a multidentate ligand that can form six bonds with a metal ion. Multidentate ligands (also called polydentate) can donate three or more lone pairs of electrons! Notice how the ligands are shaped like crabs? We call these types of ligands chelating agents. These ligands form complexes called chelates, meaning ‘crab-like’. The two molecules below, 1,2-diaminoethane (or ‘en’ for short) and the ethanedioate ion are bidentate ligands. Another thing you might notice is how the oxidation state of the ion determines the overall charge of the complex.īidentate ligands form two coordinate bonds, meaning they donate two lone pairs of electrons. Notice how the neutral ligands form positive complex ions whilst negative ligands form negative complexes. Water, chloride ions and ammonia molecules are examples of unidentate ligands.Ĭomplex ions with unidentate ligands, chemiguide and StudySmarter Unidentate ligands donate one lone pair of electrons. We classify ligands by how many coordinate bonds they can form. Reaction Quotient and Le Chatelier's Principleīefore we consider the shape of complex ions, let us briefly discuss the different types of ligands.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution.
